Nicolas Fedrigo
Age 17 | Victoria, British Columbia
CWSF Senior Gold Medal | CWSF Platinum Award for Best Senior Project | EUCYS First Place Winner | Unviersity of Toronto Engineering Award
Brian Oakley, a young father and construction worker from Victoria, BC in Canada sustained a spinal fracture while working on a job site. He was unloading a trailer when the truck shifted, causing nearly 300 kilograms of glass to fall on him, pinning him to the ground. Oakley was rushed to Victoria General Hospital and later transferred to Vancouver General Hospital for a spinal fusion by a surgeon at the top of his field. He survived the surgery, but despite excellent response times and the surgeon’s best efforts, the young father was paralyzed from the neck down due to a vertebral breach – accidental damage to the spinal cord – that occurred during the surgical procedure. This outcome is far too common, primarily caused by a rudimentary tool – the pedicle probe – used during spinal fusions (see Figure 1).
Figure 1. The traditional pedicle probe (Life Instruments, 2018).
During spinal fusion surgery, pedicle screws (a type of bone screw) are attached to the vertebrae above and below the affected vertebrae. A metal rod is then connected to the screws, stabilizing the spine. The pedicle probe is a device used by surgeons to create pilot holes that guide placement of the pedicle screws. Unfortunately, on average, 29% of patients suffer from vertebral breaches (Smith ZA, Sugimoto K, Lawton CD, & Fessler RG, 2018) from improper placement of pedicle screws, resulting in postoperative complications, such as motor defects, infection, and in many cases, paralysis.
BACKGROUND
Current approaches to address the issue of vertebral breaches during spinal fusion include electrical conductive-based navigation and fluoroscopy-based navigation, which still result in vertebral breach rates as high as 23% (Smith ZA, Sugimoto K, Lawton CD, & Fessler RG, 2018).
My goal was to make spinal fusions safer. If I redesign the pedicle probe to provide surgeons with instantaneous feedback on the probe’s location through monitoring density, this would enable them to more accurately place pedicle screws and allow for the prevention of breaches. The electromechanical pedicle probe I developed detects the difference between the higher density cortical (compact) bone and lower density cancellous (spongy) bone found in vertebrae (see Figure 2). It provides real-time feedback to surgeons, similar to the popular 1960s game “Operation”, warning of a potential vertebral breach, enabling them to redirect the probe and advance down the optimum path. This new technology is a proof-of-concept prototype that demonstrates it is possible to prevent vertebral breaches, making for more reliable spinal fusion procedures through guided trajectory navigation.
Figure 2. Higher density cortical (compact) bone forms the exterior of the vertebra and surrounds the spinal cord. An accurately placed pedicle screw remains in the lower density cancellous (spongy) bone within the vertebra. When cortical bone is cannulated (using the probe to create a pilot hole) it results in lateral or medial breaches (SpineGuard, 2018).
DESIGN AND IMPLEMENTATION
The electromechanical pedicle probe (see Figure 3) prevents vertebral breaches using density gradient analysis of cortical and cancellous bone. Cortical bone is detected by monitoring the increased cannulation force, which is a key indicator of the specific tissue type. Within the probe, applied forces are directed to a sensor tensile load cell and its voltage is scaled using an operational amplifier monitored by an Arduino Uno Rev3 microcontroller. Initially, the supporting circuit was tested on a breadboard. Once the design and testing were completed, it was reproduced and soldered onto a perfboard. After determining the magnitude of force required to cause a medial and lateral breach through the higher-density cortical bone, I developed code to warn the user when the applied force exceeds 60 Newtons (Blair-Pattison A, Hu RW, Swamy G, & Anglin C, 2018). Real-time tactile and visual feedback are achieved with an external LED and a 1000 RPM vibration motor. Additionally, the applied force is shown in real-time on a liquid-crystal display (LCD).
Figure 3. The improved pedicle probe.
The prototype pedicle probe housing was turned from Delrin, which has high stiffness, low friction, and excellent dimensional stability. The housing’s exterior was shaped ergonomically using a 3/4-inch (1.9 cm) router bit and the inside was hollowed to snugly house each of the electronic components; channels for
the wiring were also incorporated. A fitting was turned to mount inside the housing to direct the force of an interchangeable steel pedicle guide pin on the sensor tensile load cell (see Figure 4).
Figure 4. Improved pedicle probe CAD diagram. Orange component is a sensor tensile load cell, red component is a 1000 RPM vibration motor.
The Arduino Uno operates on C++ code that I wrote to accommodate the normal range of densities for both cancellous and cortical bone. However, the program also allows for easy calibration of the density threshold based on sample densities from patients with complications, which enables highly personalized procedures.
A cannulation force threshold of 60 Newtons is set for a standard patient, although personalized values can be set for those with complications such as osteoporosis. People with osteoporosis are particularly prone to fractures, meaning that they are a high-risk group for spinal fusions. This often makes these patients ineligible for this treatment, despite being in higher need of it.
As a test of accuracy and precision, ten trials of a range of nine different masses were placed onto the probe to compare the applied mass to the value displayed on the LCD.
Trials to simulate actual use were conducted on lamb cervical and lumbar vertebrae obtained from a butcher. Lamb vertebrae were selected as they closely resemble human vertebrae. Cannulation force – the force required to insert the probe – was monitored on the LCD. Bone density was calculated using the mass of tissue samples from the lamb vertebrae used in the trial divided by the corresponding volumes obtained using water displacement. The cannulation-force-to-density ratio for lamb vertebrae was compared to the accepted ratio for human vertebrae to assess the applicability of the new device to humans. Similar ratios would indicate a high likelihood of compatibility.
RESULTS
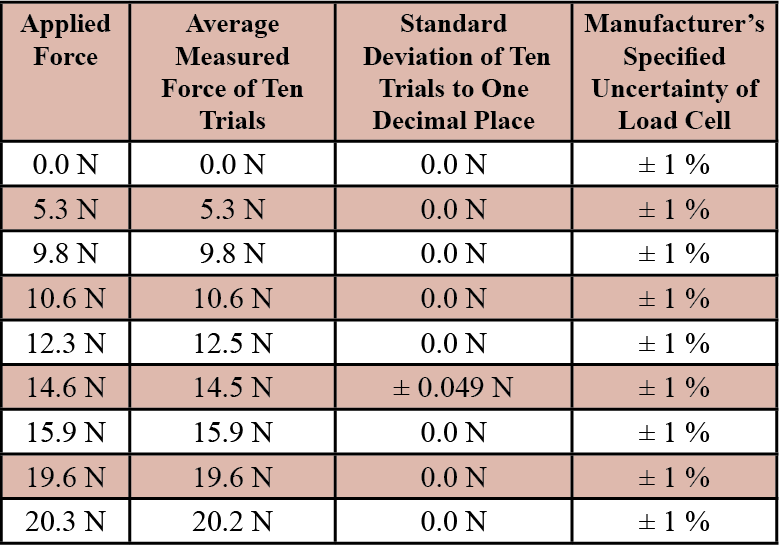
The prototype electromechanical pedicle probe was tested using nine different magnitudes of applied force, each replicated ten times. The measured values on the LCD were accurate within 0.2 Newtons of the accepted value for each applied force with an uncertainty of ±1% based on the manufacturer’s specifications of the sensor tensile load cell. The 14.6 Newton trials had a standard deviation of ±0.049 Newtons, while the other trials had a standard deviation of 0.0 Newtons since the display is limited to one decimal place (see Table 1).
Table 1. Accuracy, Standard Deviation, and Uncertainty of Improved Pedicle Probe to Applied Forces
The new probe was successful in warning the user at a preset force value. When tested on lamb vertebrae, the probe gave sufficient feedback to prevent both medial and lateral breaches. The average cannulation force was 45.7 Newtons, and the calculated cancellous tissue density was 1.05 g/cm3. The cannulation-force-to-density ratio – calculated as the cannulation force divided by the bone density – of lamb vertebrae was 43.5 (N•cm3)/g. The accepted value for humans is 44.4 (N•cm3)/g.
DISCUSSION
The similarity of the values of the cannulation-force-to-density ratio suggests that the new probe would function effectively on human vertebrae. Furthermore, the results based on the probe’s accuracy and precision demonstrate that the probe is able to consistently measure force with more than sufficient in order to detect the difference between cancellous and cortical bone.
This novel approach of guiding the surgeon through density gradient analysis keeps procedure and feedback delay to a fraction of other approaches, while eliminating potentially harmful ionizing radiation exposure for surgeons, patients, and hospital staff. Furthermore, it does not depend on, or require, any preoperative imaging.
The cost per unit of manufacturing the improved pedicle probe is less than $56 CAD ($42 USD), which could be significantly reduced if components were purchased in bulk. Other alternatives such as fluoroscopy imaging systems cost over $133,815 CAD ($100,000 USD) per unit with additional expenses for various attachments.
Finally, the redesigned probe maintains a sense of familiarity for surgeons used to the traditional approach by retaining the size and form of the standard probe. This increases the likelihood of adoption and eliminates the need for significant retraining on the procedure.
FUTURE WORK
The new electromechanical probe could be used differently for other procedures and has the potential to be improved. I am inspired to enhance its functionality by developing a Bluetooth wireless version, which has been accounted for in the current design. The probe would still have an external control board; however, it would not be connected to the probe itself. I would also like to arrange for testing of the new probe on human vertebrae or cadavers. Ultimately, I am hopeful that the device will be used in human clinical trials.
I am currently working on the development of an enhancement that would actively prevent the penetration of cortical bone regardless of the surgeon’s actions. My ultimate goal is for the new probe to significantly reduce, and ideally, eliminate human error in spinal fusion surgeries and related procedures.
CONCLUSIONS
The redesigned pedicle probe measures cannulation force with sufficient accuracy to pass as desired through cancellous bone, but detect and warn the user with visual and tactile feedback when it encounters cortical bone. Enhancing a surgeon’s ability to determine an appropriate path for pedicle screws through a sensor-enabled probe has the potential to significantly reduce the incidence of vertebral breaches during spinal fusion surgery. Using this new technology, personalized procedures could be implemented through custom code and specific calibration, significantly reducing the risk for those with complications.
Results of patent searches for Canada, the United States, and Europe suggest that the redesigned probe is unique in predicting and preventing breaches in spinal fusions based on predetermined force threshold values. The probe is also unique in the sense of enabling personalized procedures in spinal fusions for those with complications, through calibrating a control (force) limit based on tissue samples prior to the procedure.
Most importantly, test results with the prototype suggest that the new probe could prevent potential vertebral breaches on human patients – a significant benefit for those who require this critical medical procedure and to the surgeons who perform them.
ACKNOWLEDGEMENTS
I would like to thank Mr. Reni Barlow of Youth Science Canada for his tremendous support. I would also like to acknowledge Mr. Sean Hayes and Mr. John Vucko of Claremont Secondary School in Victoria, British Columbia, Canada for their support throughout my experiences in science fairs and inspiring me to compete in the Vancouver Island Regional Science Fair three years ago with my first innovation
REFERENCES
Smith, Z., Sugimoto, K., Lawton, C., & Fessler, R. (2014). Incidence of Lumbar Spine Pedicle Breach After Percutaneous Screw Fixation. Journal Of Spinal Disorders And Techniques, 27(7), 358-363. doi: 10.1097/bsd.0b013e31826226cb
Brasiliense, L., Theodore, N., Lazaro, B., Sayed, Z., Deniz, F., Sonntag, V., & Crawford, N. (2010). Quantitative analysis of misplaced pedicle screws in the thoracic spine: how much pullout strength is lost?. Journal Of Neurosurgery: Spine, 503-508. doi: 10.3171/2009.11.spine09408
Blair-Pattison, A., Hu, R., Swamy, G., & Anglin, C. (2014). Forces in spinal cannulation and breaches ex vivo. Proceedings Of The Institution Of Mechanical Engineers, Part H: Journal Of Engineering In Medicine, 228(7), 693-702. doi: 10.1177/0954411914540632
SpineGuard - Patients - Spinal Fusion Overview. (2019). Retrieved from http://www.spineguard.com/patients-spinal-fusion/spinal-fusion-overview/
Probe, P. (2019). Pedicle Probe | lifeinstruments.com. Retrieved from http://lifeinstruments.com/product/pedicle-probe/
BIBLIOGRAPHY
Sheng, S., Wang, X., Xu, H., Zhu, G., & Zhou, Y. (2009). Anatomy of large animal spines and its comparison to the human spine: a systematic review. European Spine Journal, 19(1), 46-56. doi: 10.1007/s00586-009-1192-5
Blanton, P., & Biggs, N. (1968). Density of fresh and embalmed human compact and cancellous bone. American Journal Of Physical Anthropology, 29(1), 39-44. doi: 10.1002/ajpa.1330290113
Victoria father paralyzed from neck down after work related accident in Metchosin - Victoria Buzz. (2019). Retrieved from http://victoriabuzz.com/2016/07/victoria-father-paralyzed-from-neck-down-after-work-related-accident-in-victoria
NICOLAS FEDRIGO
I am a Grade 12 student from Claremont Secondary School and I have competed in the Canada-Wide Science Fair for the past three years. This year I was honoured to receive the Platinum Award in the senior division. My inspiration for this year’s project is that up twenty-nine percent of patients who undergo spinal fusions suffer from vertebral breaches which cause complications such as infection and paralysis. This inspired me to make spinal fusions safer through redesigning the pedicle probe. My experiences in science fairs taught me about the field of biomedical engineering and I am inspired to pursue a career in this field. In my spare time, I am the president and founder of the Claremont Secondary Science and Engineering Club, along with other volunteering experiences. I have also coached as a swim instructor for elementary school children at a local recreation centre for over three years now. Additionally, I am a research intern at the Deeley Research Centre - part of the BC Cancer Agency.